Sous-titre :
How dual-material engineering, rigorous YBB/ISO testing, and GMP-compliant manufacturing make composite caps the trusted choice for injectables, vaccines, and infusions worldwide.
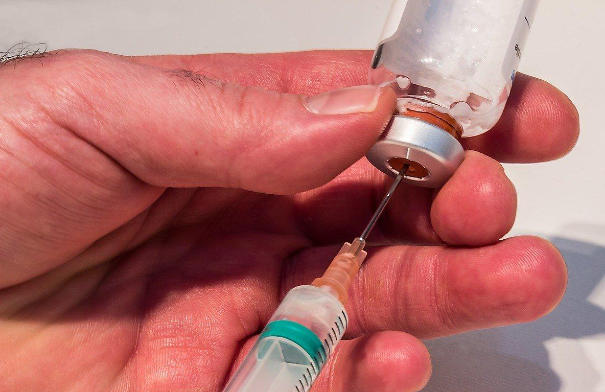
In the high-stakes world of pharmaceutical packaging, every component must perform flawlessly — from raw material selection to final sterilization. Among the most critical yet often overlooked elements is the closure system. For parenteral products like infusions, vaccines, and biologics, the capuchon combiné aluminium-plastique has emerged as the industry’s “reliable guardian” — delivering unmatched sealing performance, mechanical resilience, and user safety.
Au PharGlass, we’ve engineered our aluminum-plastic caps based on decades of R&D, real-world feedback, and strict adherence to Chinese Pharmacopoeia (YBB) and international standards. In this technical deep-dive, we break down pourquoi these composite caps are indispensable for modern drug delivery — backed by material science, validated test methods, and global regulatory alignment.
🔧 1. Dual-Layer Architecture: Strength Meets Flexibility
The secret behind the aluminum-plastic cap’s reliability lies in its two-component design:
🛡️ Aluminum Outer Shell – The Protective Barrier
- Function: Provides structural rigidity, corrosion resistance, and impermeability against oxygen, moisture, and light.
- Design Feature: Pre-scored tear-off ring or flip-tab enables tool-free, one-handed opening — crucial in clinical settings.
- Material: Typically alloy 8011 or 8079, anodized or lacquered for enhanced durability.
💧 Plastic Inner Liner – The Sealing Engine
- Function: Creates a hermetic seal via elastic compression against the vial neck.
- Materials: Medical-grade Polyéthylène (PE) ou Polypropylène (PP) — selected for chemical inertness, thermal stability, and low extractables.
- Compatibilité : Works seamlessly with glass, plastic, or coated bottles across multiple pharmacopoeias.
✅ Industry Insight: According to USP <670>, primary closures must maintain integrity under stress conditions including temperature cycling, vibration, and drop tests. Our caps exceed these requirements through optimized wall thickness and precision molding tolerances.
⚙️ 2. Performance Advantages: Why Pharma Trusts Composite Caps
Our aluminum-plastic caps deliver four core benefits that directly impact drug safety, operational efficiency, and patient compliance:
1. Superior Seal Integrity
The rigid aluminum shell + elastic plastic liner creates a dual-mechanism seal:
- Mechanical interlock during crimping
- Elastic rebound after sterilization → zero leakage even after thermal cycling
Validated via vacuum decay method (leak rate ≤ 1×10⁻⁶ mbar·L/s) and microbial challenge post-autoclaving.
2. Ergonomic & Tamper-Evident Opening
Pre-engineered tear points ensure consistent opening force (8–15 N), while irreversible breakage provides visual tamper evidence — meeting FDA 21 CFR Part 211 and EU Annex 1 requirements.
3. Corrosion & Chemical Resistance
Both aluminum and PP/PE exhibit excellent resistance to acids, bases, alcohols, and saline solutions — making them suitable for virtually all liquid formulations, from antibiotics to biologics.
4. Long-Term Stability
Unlike pure plastic caps prone to creep or deformation, our composite structure maintains dimensional stability over time — ensuring consistent seal performance throughout shelf life (up to 5 years).
🧪 3. Rigorous Testing Protocol: Validated Against YBB & Global Standards
Quality isn’t assumed — it’s proven. Our caps undergo comprehensive testing per YBB 00402003-2015 et YBB 00372003-2015, which align closely with ISO 15378 and ASTM D3475 guidelines:
✔️ Aluminum Material Mechanical Properties
- Tensile Strength & Elongation Test: Ensures caps withstand capping pressure, transport shocks, and autoclave cycles without cracking or delaminating.
- Acceptance Criteria: Minimum tensile strength ≥ 80 MPa; elongation ≥ 15%.
✔️ Opening Force Test
- Measures torque required to remove cap using standardized pull-test apparatus.
- Target Range: 8–15 Newtons for adult use; 5–8 N for pediatric applications.
- Ensures balance between security and usability.
✔️ Plastic Removal Force Test
- Evaluates force needed to detach plastic liner from aluminum shell during opening.
- Prevents partial detachment or residue left on vial neck — critical for sterile compounding.
✔️ Tear-Off Strip Force Test (ZD/OD Type)
- Simulates real-world opening scenario until complete removal.
- Validates consistency of tear initiation and propagation — no jagged edges or incomplete breaks.
📊 Conseil de pro : We also offer optional E&L (Extractables & Leachables) studies per BPOG protocol and biocompatibility testing per ISO 10993-1 for sensitive biologics.
🏭 4. GMP-Compliant Manufacturing: From Coil to Carton
Producing medical-grade aluminum-plastic caps requires micron-level control across every stage:
✔️ Raw Material Selection
- Aluminum coils degreased and rinsed with ionized water before stamping
- PP/PE resin USP Class VI certified, low endotoxin (<0.25 EU/mL)
✔️ Cleanroom Assembly
- Post-washing components enter ISO Class 8 (Grade D) environment
- Fully automated conveyors eliminate manual handling
- Local Grade A laminar flow during final assembly and packaging
✔️ Final QC & Traceability
- Dimensional accuracy: ±0.05mm tolerance on inner diameter
- Visual inspection: AI-powered cameras detect scratches, dents, or incomplete scoring
- Batch traceability via MES system — full documentation available upon request (CoA, DMF, Sterilization Certificate)
🌍 5. Global Applications & Customization Options
Our caps support diverse pharmaceutical formats:
| Type de produit | Taille de bouchon recommandée | Dossier spécial |
|---|---|---|
| Infusion Solutions | 20 mm | High-temp sterilization ready |
| Vaccines | 13mm / 15mm | Low-extractable liner option |
| Biologics | 20 mm | Gamma-radiation compatible |
| Pediatric Syrups | 15 mm | Child-resistant variant available |
Nous proposons également des services de personnalisation :
- Embossage du logo / marquage au laser
- Couleurs personnalisées (or, argent, bleu, vert, rouge)
- Géométrie modifiée de la gaine pour les formulations visqueuses
- Documentation soutenue par la DMF pour les soumissions américaines/européennes
📈 6. Sustainability & Regulatory Alignment
Au-delà de la fonctionnalité, les acheteurs modernes exigent une responsabilité environnementale :
- ♻️ Matériaux recyclables: L'aluminium est recyclable à l'infini ; le PP peut être séparé et traité dans des installations à flux mixte.
- 🌱 Conception légère: Reduces carbon footprint during logistics (~5–8g per unit vs. 10–15g for all-metal alternatives).
- 📜 Appui réglementaire: Nous fournissons sur demande des rapports CoA, E&L, des certificats de stérilisation et des références DMF.
Nos installations font l'objet d'un audit annuel par des tiers pour vérifier la conformité aux normes ISO 15378, ISO 9001 et SEDEX, ce qui vous donne la garantie d'un approvisionnement éthique et d'une qualité constante.
✅ Conclusion: More Than Just a Cap — It’s a Safety System
From barrier protection to ergonomic usability, from sterilization resilience to cost savings — aluminum-plastic combination caps represent the optimal solution for today’s dynamic pharmaceutical market.
Au PharGlass, we don’t just manufacture caps — we engineer partnerships. Whether you’re launching a new biologic, scaling vaccine distribution, or optimizing legacy injectable formats, our team stands ready to tailor every parameter — from alloy composition to color coding — to meet your unique needs.
📩 Contactez-nous dès aujourd'hui pour demander des échantillons, des fiches techniques ou programmer une visite virtuelle de l'usine.