

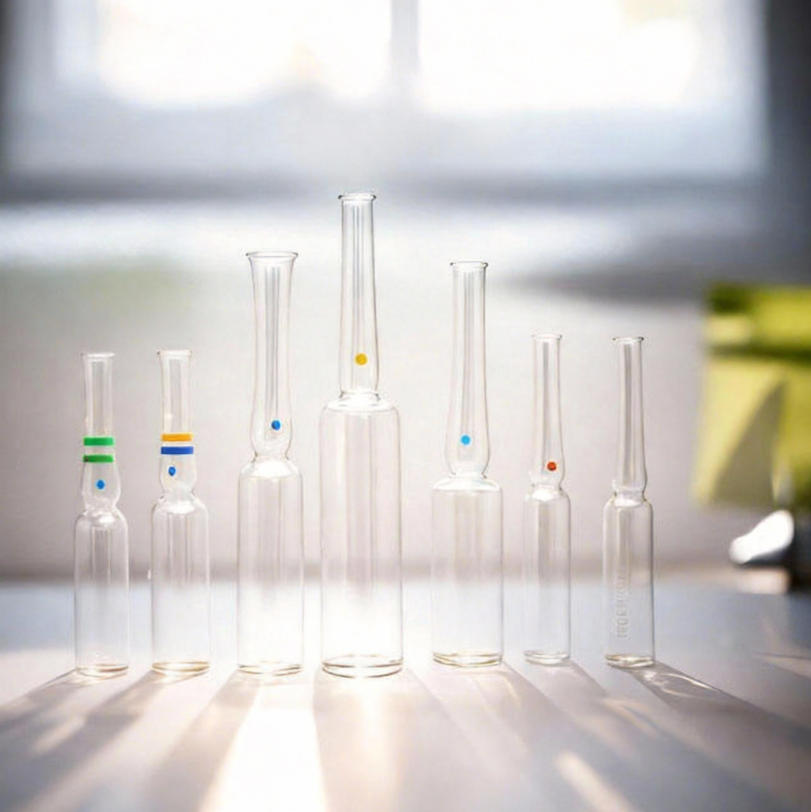

Borosilicate Glass Ampoules ISO Injectable 1ml–20ml OPC: Where Precision Engineering Meets Visual Integrity in Pharmaceutical Packaging
Premium Type I Borosilicate Glass Ampoules – Designed for High-Purity Injectables, Biologics, and Luxury Skincare Applications with ISO Compliance et OPC Certification
The Borosilicate Glass Ampoule ISO Injectable Pharmaceutical 1ml 2ml 5ml 10ml 20ml OPC Glass Ampoule from PharGlass represents a new standard in primary packaging excellence—where chemical inertness, thermal resilience, and aesthetic precision converge.
Manufactured under ISO 9001:2015 et GMP-compliant conditions in Henan, China, these medical-grade glass ampoules are crafted from neutral borosilicate glass (Type I) to meet the most stringent requirements of USP, EP 3.1.4, YBB 0032-2015, and ISO 4803 standards. Available in clear or amber finishes, with gold-stamped labeling, they are ideal for single-dose injectables, vaccines, biologic therapies, and even high-end cosmetic serums.
Notre OPC-certified production process ensures zero contamination, consistent wall thickness (0.5 cm), and ultra-low particulate levels—critical for both drug stability et patient safety.
🎯 Why Borosilicate Glass Is the Gold Standard for Parenteral Products
Unlike soda-lime or low-borosilicate glass, borosilicate glass contains over 8% boron oxide (B₂O₃), which dramatically improves:
- ✅ Hydrolytic resistance — reduces alkali leaching by up to 90%
- ✅ Thermal shock resistance — withstands autoclaving at 121°C without cracking
- ✅ Chemical inertness — minimizes interaction with sensitive formulations
According to a 2022 study published in Journal of Pharmaceutical Sciences, Type I borosilicate glass reduced protein aggregation in monoclonal antibody solutions by 74% compared to Type II glass over 12 months of storage.
This is why regulatory agencies worldwide mandate its use for parenteral products that are:
- pH-sensitive
- chemically reactive
- high-value biologics
🔍 The Role of OPC Certification in Modern Pharma Packaging
OPC (One Piece Control) certification is not just a quality mark—it’s a process philosophy that ensures every single ampoule is traceable, inspected, and validated before shipment.
Au PharGlass, our OPC-certified line includes:
- Automated visual inspection (AI-powered defect detection)
- Particle count testing (<50 particles/mL per ISO 11137)
- Wall thickness uniformity (±0.05 mm across all sizes)
- Batch-level tracking from raw material to final carton
This level of control is essential for FDA inspections, EU GMP audits, and global market access.
🖼️ Design & Aesthetics: From Function to Brand Identity
While performance is paramount, visual design plays a crucial role in brand perception et market differentiation.
Notre Borosilicate Glass Ampoules feature:
- Elegant neck-to-body ratio for smooth break-off
- Gold hot stamping for premium branding (customizable font, logo, batch code)
- Optional amber tint for UV protection of light-sensitive drugs (e.g., tetracyclines, nitrates)
- Clear glass for visibility during inspection (ideal for vaccines)
In the luxury skincare industry, brands like SK-II, La Prairie, and Drunk Elephant use amber borosilicate ampoules to signal purity, potency, and premium formulation—even when the product isn’t injectable.
🌐 Global Market Compatibility: From USP to YBB to ISO
| Standard | Key Requirement | PharGlass Compliance |
|---|---|---|
| USP | Type I glass required for parenterals | ✔️ Full compliance |
| EP 3.1.4 | Hydrolytic resistance test (Class I) | ✔️ Passes Class I |
| YBB 0032-2015 | Chinese pharmacopeial standard for glass containers | ✔️ Certified |
| ISO 4803 | International standard for glass ampoules | ✔️ Compliant |
This multi-standard alignment allows your product to be registered in North America, Europe, China, and ASEAN markets without reformulation.
💡 Size Matters: How Capacity Affects Stability & Handling
| Volume | Typical Use Case | Critical Considerations |
|---|---|---|
| 1ml–3ml | Biologics, peptides, high-concentration drugs | Higher surface-area-to-volume ratio → greater leachables risk |
| 5ml–10ml | Antibiotics, vaccines, general injectables | Balanced cost-performance |
| 20ml | High-volume injectables, IV solutions | Requires robust sealing and thicker walls |
All sizes are produced on dedicated lines to prevent cross-contamination and ensure consistent quality.
✅ Why Choose PharGlass for Your Borosilicate Ampoules?
- 🔬 Full DMF support for FDA submissions
- 📄 COA, TDS, and extractables data available upon request
- 🧪 Custom E&L studies for complex formulations
- 🛠️ OEM/ODM services: custom size, color, printing, and neck geometry
- 🚚 Global shipping with DDP/DAP options
- 🧩 Free samples for compatibility testing
We also offer pre-sterilized versions (gamma or autoclave), tamper-evident seals, and custom label stickers.
📞 Need Help Selecting the Right Ampoule?
Our packaging engineers can review your formulation and recommend the optimal glass type, size, and finish—free of charge.
Contact us at sales@pharglass.com or visit https://www.pharglass.com.


