Untertitel:
How dual-material engineering, rigorous YBB/ISO testing, and GMP-compliant manufacturing make composite caps the trusted choice for injectables, vaccines, and infusions worldwide.
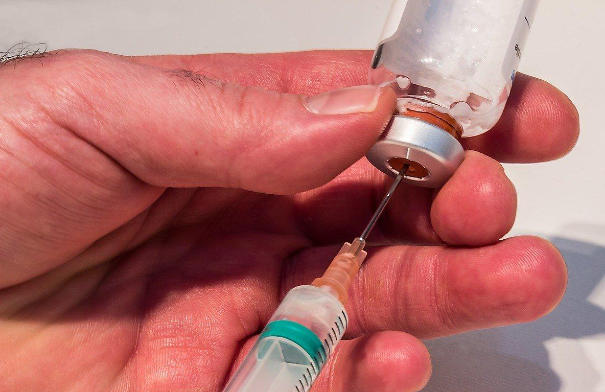
In the high-stakes world of pharmaceutical packaging, every component must perform flawlessly — from raw material selection to final sterilization. Among the most critical yet often overlooked elements is the closure system. For parenteral products like infusions, vaccines, and biologics, the aluminum-plastic combination cap has emerged as the industry’s “reliable guardian” — delivering unmatched sealing performance, mechanical resilience, and user safety.
Unter PharGlass, we’ve engineered our aluminum-plastic caps based on decades of R&D, real-world feedback, and strict adherence to Chinese Pharmacopoeia (YBB) and international standards. In this technical deep-dive, we break down warum these composite caps are indispensable for modern drug delivery — backed by material science, validated test methods, and global regulatory alignment.
🔧 1. Dual-Layer Architecture: Strength Meets Flexibility
The secret behind the aluminum-plastic cap’s reliability lies in its two-component design:
🛡️ Aluminum Outer Shell – The Protective Barrier
- Funktion: Provides structural rigidity, corrosion resistance, and impermeability against oxygen, moisture, and light.
- Design-Merkmal: Pre-scored tear-off ring or flip-tab enables tool-free, one-handed opening — crucial in clinical settings.
- Material: Typically alloy 8011 or 8079, anodized or lacquered for enhanced durability.
💧 Plastic Inner Liner – The Sealing Engine
- Funktion: Creates a hermetic seal via elastic compression against the vial neck.
- Materials: Medical-grade Polyethylen (PE) oder Polypropylen (PP) — selected for chemical inertness, thermal stability, and low extractables.
- Kompatibilität: Works seamlessly with glass, plastic, or coated bottles across multiple pharmacopoeias.
✅ Einblick in die Industrie: According to USP <670>, primary closures must maintain integrity under stress conditions including temperature cycling, vibration, and drop tests. Our caps exceed these requirements through optimized wall thickness and precision molding tolerances.
⚙️ 2. Performance Advantages: Why Pharma Trusts Composite Caps
Our aluminum-plastic caps deliver four core benefits that directly impact drug safety, operational efficiency, and patient compliance:
1. Superior Seal Integrity
The rigid aluminum shell + elastic plastic liner creates a dual-mechanism seal:
- Mechanical interlock during crimping
- Elastic rebound after sterilization → zero leakage even after thermal cycling
Validated via vacuum decay method (leak rate ≤ 1×10⁻⁶ mbar·L/s) and microbial challenge post-autoclaving.
2. Ergonomic & Tamper-Evident Opening
Pre-engineered tear points ensure consistent opening force (8–15 N), while irreversible breakage provides visual tamper evidence — meeting FDA 21 CFR Part 211 and EU Annex 1 requirements.
3. Corrosion & Chemical Resistance
Both aluminum and PP/PE exhibit excellent resistance to acids, bases, alcohols, and saline solutions — making them suitable for virtually all liquid formulations, from antibiotics to biologics.
4. Long-Term Stability
Unlike pure plastic caps prone to creep or deformation, our composite structure maintains dimensional stability over time — ensuring consistent seal performance throughout shelf life (up to 5 years).
🧪 3. Rigorous Testing Protocol: Validated Against YBB & Global Standards
Quality isn’t assumed — it’s proven. Our caps undergo comprehensive testing per YBB 00402003-2015 und YBB 00372003-2015, which align closely with ISO 15378 and ASTM D3475 guidelines:
✔️ Aluminum Material Mechanical Properties
- Tensile Strength & Elongation Test: Ensures caps withstand capping pressure, transport shocks, and autoclave cycles without cracking or delaminating.
- Acceptance Criteria: Minimum tensile strength ≥ 80 MPa; elongation ≥ 15%.
✔️ Opening Force Test
- Measures torque required to remove cap using standardized pull-test apparatus.
- Target Range: 8–15 Newtons for adult use; 5–8 N for pediatric applications.
- Ensures balance between security and usability.
✔️ Plastic Removal Force Test
- Evaluates force needed to detach plastic liner from aluminum shell during opening.
- Prevents partial detachment or residue left on vial neck — critical for sterile compounding.
✔️ Tear-Off Strip Force Test (ZD/OD Type)
- Simulates real-world opening scenario until complete removal.
- Validates consistency of tear initiation and propagation — no jagged edges or incomplete breaks.
📊 Profi-Tipp: We also offer optional E&L (Extractables & Leachables) studies per BPOG protocol and biocompatibility testing per ISO 10993-1 for sensitive biologics.
🏭 4. GMP-Compliant Manufacturing: From Coil to Carton
Producing medical-grade aluminum-plastic caps requires micron-level control across every stage:
✔️ Auswahl der Rohmaterialien
- Aluminum coils degreased and rinsed with ionized water before stamping
- PP/PE resin USP Class VI certified, low endotoxin (<0.25 EU/mL)
✔️ Reinraum Montage
- Post-washing components enter ISO Class 8 (Grade D) environment
- Fully automated conveyors eliminate manual handling
- Local Grade A laminar flow during final assembly and packaging
✔️ Final QC & Traceability
- Maßgenauigkeit: ±0,05 mm Toleranz am Innendurchmesser
- Visuelle Inspektion: KI-gestützte Kameras erkennen Kratzer, Dellen oder unvollständige Rillen
- Batch traceability via MES system — full documentation available upon request (CoA, DMF, Sterilization Certificate)
🌍 5. Global Applications & Customization Options
Our caps support diverse pharmaceutical formats:
| Produkttyp | Empfohlene Kappengröße | Besonderes Merkmal |
|---|---|---|
| Infusion Solutions | 20mm | High-temp sterilization ready |
| Vaccines | 13mm / 15mm | Low-extractable liner option |
| Biologics | 20mm | Gamma-radiation compatible |
| Pediatric Syrups | 15mm | Child-resistant variant available |
Wir bieten auch Anpassungsdienste an:
- Logo-Prägung / Lasermarkierung
- Benutzerdefinierte Farben (Gold, Silber, Blau, Grün, Rot)
- Geänderte Geometrie des Liners für viskose Formulierungen
- DMF-unterstützte Dokumentation für US/EU-Einreichungen
📈 6. Sustainability & Regulatory Alignment
Über die Funktionalität hinaus verlangen die modernen Käufer Umweltverantwortung:
- ♻️ Recycelbare Materialien: Aluminium ist unbegrenzt recycelbar; PP kann getrennt und in Mischstromanlagen verarbeitet werden.
- 🌱 Leichte Konstruktion: Reduces carbon footprint during logistics (~5–8g per unit vs. 10–15g for all-metal alternatives).
- 📜 Regulatorische Unterstützung: Wir stellen auf Anfrage CoA, E&L-Berichte, Sterilisationszertifikate und DMF-Referenzen zur Verfügung.
Unsere Einrichtungen werden jährlich von Dritten auf die Einhaltung der Normen ISO 15378, ISO 9001 und SEDEX geprüft - so können Sie sich auf eine ethische Beschaffung und gleichbleibende Qualität verlassen.
✅ Conclusion: More Than Just a Cap — It’s a Safety System
From barrier protection to ergonomic usability, from sterilization resilience to cost savings — aluminum-plastic combination caps represent the optimal solution for today’s dynamic pharmaceutical market.
Unter PharGlass, we don’t just manufacture caps — we engineer partnerships. Whether you’re launching a new biologic, scaling vaccine distribution, or optimizing legacy injectable formats, our team stands ready to tailor every parameter — from alloy composition to color coding — to meet your unique needs.
📩 Kontaktieren Sie uns heute um Muster und technische Datenblätter anzufordern oder eine virtuelle Werksbesichtigung zu vereinbaren.