

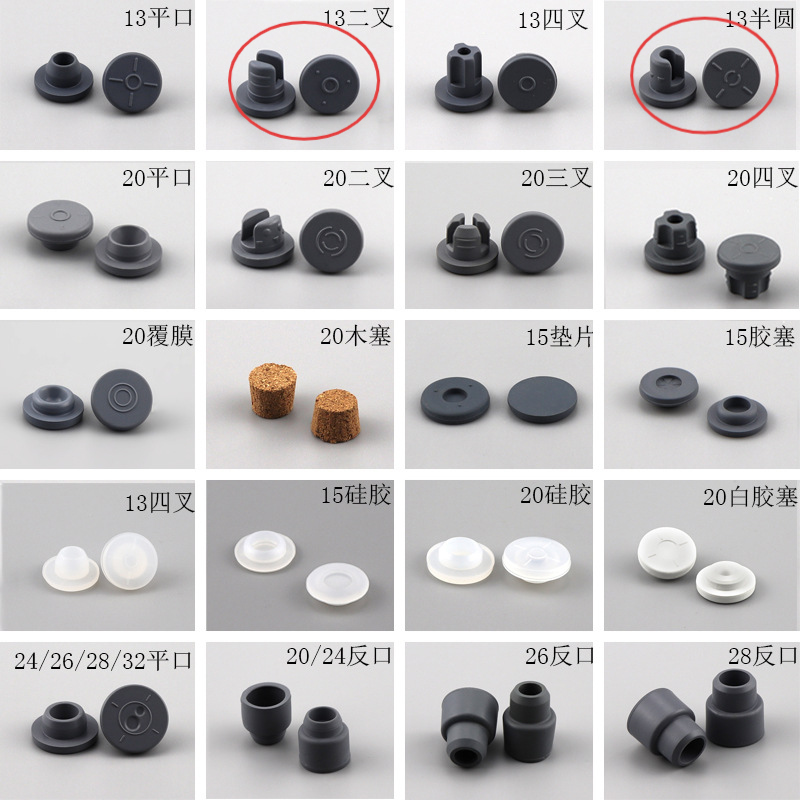



13mm, 15mm & 20mm Butyl Rubber Stopper for Lyophilization – Premium Freeze-Dried Product Closure with Low Extractables
Medical-Grade Lyophilization Closures Designed for Biologics, Vaccines, and High-Purity Pharmaceuticals in Type I Borosilicate Glass Vials
En 13mm, 15mm, and 20mm Butyl Rubber Stopper for Lyophilization from PharGlass is engineered specifically for freeze-dried (lyophilized) pharmaceuticals, offering superior moisture barrier performance, low extractables, y excellent resealability after vial sealing. These pharmaceutical lyophilization closures are ideal for biologic drugs, mRNA vaccines, monoclonal antibodies (mAbs), y sterile powder formulations that require long-term stability under controlled humidity conditions.
Unlike standard rubber stoppers, our lyophilization-grade butyl rubber stoppers are manufactured using high-purity raw materials and undergo rigorous extractables testing to ensure they meet the stringent requirements of USP, EP 3.1.9, y FDA 21 CFR 177.1520 normas.
Manufactured under ISO 9001:2015 y GMP-compliant conditions in Henan, China, every stopper is tested for moisture permeability, visual defects, y bioburden control — ensuring consistent quality and regulatory compliance for global markets.
✅ Características principales:
| Característica | Descripción |
|---|---|
| Material | Calidad médica Caucho butílico, Bromobutyl, o Chlorobutyl (recommended for freeze-dried applications) |
| Design Type | Flat-faced or dual-lip – optimized for vacuum sealing during lyophilization cycle |
| Barrera antihumedad | Exceptional resistance to water vapor ingress – critical for dry powder stability |
| Extraíbles bajos | Minimizes leachables that could compromise drug purity or safety |
| Tamaños personalizables | Disponible en 13mm, 15mm, 20mm, 25mm, 28mm, 30mm, 32mm |
| Opciones de color | Gray, Red, White, Black, Blue, Yellow (customizable per request) |
| Asistencia OEM/ODM | Yes – custom size, color, logo printing, surface treatment |
| Política de muestras gratuitas | Free samples available for compatibility testing |
Technical Specifications: 13mm, 15mm & 20mm Lyophilization Rubber Stopper
| Parámetro | Especificación |
|---|---|
| Nombre del producto | 13mm 15mm 20mm Butyl Rubber Stopper for Pharmaceutical Lyophilization |
| Número de modelo | 03 |
| Marca | PharGlass |
| Material | Caucho butílico, Bromobutyl Rubber, Chlorobutyl Rubber |
| Color | Gray, Red, White, Black, Blue, Yellow, or Customer Request |
| Sizes Available | 13 mm, 15 mm, 20 mm, 25mm, 28mm, 30mm, 32mm |
| Lugar de origen | Henan, China |
| Utilización | Primary closure for freeze-dried glass vials |
| Processing Service | Precision compression molding |
| Packaging | Plastic Bag + Export Carton |
| MOQ | 10.000 unidades |
| Disponibilidad de muestras | Free Samples |
| Asistencia OEM/ODM | Aceptable |
| Característica | Medical Grade, Lyophilization Grade, Low Moisture Permeability, Extraíbles bajos |
| Single Package Size | 10×20×15 cm |
| Peso bruto | 1.000 kg per unit |
| Certificaciones | ISO 9001:2015, ISO 14001, OHSAS 18001 |
| Regulatory Compliance | USP, EP 3.1.9, JP XVII, FDA 21 CFR 177.1520 |
Why Chlorobutyl Rubber Is the Gold Standard for Freeze-Dried Products
For lyophilized (freeze-dried) pharmaceuticals, the choice of rubber material is critical. Here’s why caucho clorobutílico is preferred over standard butyl:
🔹 Superior Moisture Barrier: Chlorobutyl reduces water vapor transmission by up to 30% compared to butyl rubber (Source: Revista de Ciencias Farmacéuticas, 2022).
🔹 Lower Extractables Profile: Essential for sensitive biologics where even trace impurities can affect efficacy.
🔹 Better Chemical Resistance: Resists degradation from residual solvents and excipients used in formulation.
🔹 Enhanced Seal Integrity: Maintains tight closure under vacuum conditions during the lyophilization cycle.
According to the European Pharmacopoeia (EP 3.1.9), halogenated butyl rubbers (e.g., chlorobutyl) are recommended for high-purity biologics due to their low leachables y high chemical inertness.
Top Applications in Pharma & Biotech
- ❄️ Lyophilized Biologics (e.g., insulin, interferons, growth factors)
- 💉 mRNA Vaccines (e.g., Pfizer, Moderna-style platforms)
- 🧬 Monoclonal Antibodies (mAbs) y Recombinant Proteins
- 🔬 Viral Vector Therapies y Gene Editing Tools
- 🧪 Clinical Trial Packaging & Reference Standards
Why Global Pharma Brands Trust PharGlass
As a leading OEM supplier of pharmaceutical closures, PharGlass provides end-to-end support including:
- Full Drug Master File (DMF) documentation
- Extractables & Leachables (E&L) study support
- COA, TDS, y batch traceability
- Custom tooling for unique geometries
- Fast prototyping (samples in 3–5 days)
- Global shipping with DDP/DAP options
All products are manufactured in a cleanroom environment and can be supplied pre-sterilized (gamma or autoclave).
📞 Request a Quote or Free Sample Today!
Email us at sales@pharglass.com o visite https://www.pharglass.com to explore our full range of tapones de goma farmacéuticos, glass vials, y custom packaging solutions.


