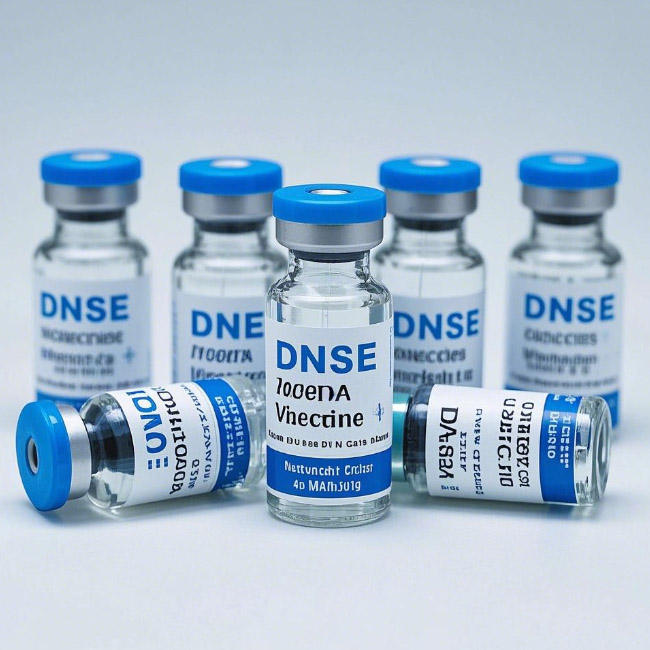
mRNA Vaccine Vials – YBB & USP Type I Compliant, 2ml–20ml, -70°C Stable, Flip-Off Seal
Ultra-Cold Resistant Primary Packaging by Pharglass | For mRNA/LNP Formulations | Ready for Global Regulatory Submissions
Pharglass mRNA vaccine vials are engineered to meet the extreme demands of next-generation nucleic acid therapeutics. Made from Type I neutral borosilicate glass and compliant with USP, فصل ۳.۲.۱, and YBB00292005-2-2015, these vials deliver unmatched chemical inertness, low extractables, and critical cryogenic stability down to -70°C—ensuring integrity during ultra-cold chain storage and transport of mRNA-LNP (lipid nanoparticle) vaccines.
Available in 2ml, 5ml, 10ml, and 20ml capacities, each vial features a color-coded flip-off seal with a bromobutyl rubber stopper, enabling easy visual identification, needle access, and resealability—ideal for multi-dose withdrawal in clinical or mass-vaccination settings.
Designed for seamless integration into aseptic fill-finish lines, Pharglass mRNA vials support both sterile (ETO/gamma) و non-sterile configurations, with full documentation for FDA, EMA, and NMPA regulatory filings.
Why These Vials Are Essential for mRNA Vaccines
mRNA formulations are highly sensitive to:
- Temperature fluctuations (requiring stable -70°C storage)
- Container surface interactions (leading to aggregation or degradation)
- Particulate contamination (triggering immune responses)
Pharglass addresses these challenges through:
- Thermal shock-resistant borosilicate glass tested through 100+ freeze-thaw cycles
- Smooth inner surface finish to minimize LNP adhesion
- Rigorous particulate control during manufacturing
- Flip-off seals that maintain closure integrity even after repeated punctures
Key Features & Benefits
- ✅ -70°C Cryogenic Stable – Validated for long-term ultra-cold storage
- ✅ USP/EP/YBB Type I Compliant – Highest pharmaceutical glass standard
- ✅ Flip-Off Seal with Bromobutyl Stopper – Easy access, tamper-evident, resealable
- ✅ Low Leachables & Low Binding – Preserves mRNA-LNP stability
- ✅ Color-Coded Caps – Supports dose differentiation (e.g., purple for 0.3ml, gray for 0.5ml*)
- ✅ Global Regulatory Ready – DMF support and full CoA available
*Cap colors customizable per client requirements.
Technical Specifications
| Parameter | Specification |
|---|---|
| Product Name | mRNA Vaccine Vials |
| Brand | Pharglass |
| Capacities | 2ml, 3ml, 5ml, 10ml, 15ml, 20ml (standard); custom sizes available |
| Glass Type | Type I Neutral Borosilicate Glass |
| Compliance | USP <660>, EP 3.2.1, YBB00292005-2-2015, ISO 8362-1 |
| Temperature Stability | -70°C to +40°C (validated for cryogenic storage) |
| Closure System | Flip-off aluminum cap + coated bromobutyl rubber stopper |
| Endotoxin Level | <0.25 EU/ml (optional; standard for sterile versions) |
| Sterility Options | Non-sterile (standard); ETO or gamma sterilized (SAL 10⁻⁶) on request |
| Packaging | Bulk in cartons, or nest-tray packed for automated filling |
| MOQ | 5,000 pieces |
| Applications | mRNA-LNP vaccines, self-amplifying RNA (saRNA), DNA vaccines, viral vectors, biologics |
Trusted by Innovators in Genetic Medicine
Pharglass mRNA vials are used by:
- Leading mRNA platform companies
- Government vaccine stockpile programs
- CDMOs supporting Phase I–III trials
- Biotech startups developing novel nucleic acid therapeutics
Our manufacturing facilities operate under ISO 15378 (pharmaceutical packaging) and ISO 13485 (medical devices) quality systems, ensuring batch-to-batch consistency and full traceability.
Customization & Support
- Pre-slit or coated stoppers for automated filling
- Unique serialization or laser marking
- Regulatory dossier support (CEP, DMF, ASMF)
- Sample kits with stability data
Engineered for the future of medicine. Validated for today’s most demanding vaccines.
When your mRNA formulation can’t afford container risk, choose Pharglass—where science, safety, and supply chain reliability converge.
📩 Contact us today for samples, stability reports, or a quote tailored to your vaccine program!