




13mm, 15mm & 20mm Butyl Rubber Stopper for Lyophilization – Premium Freeze-Dried Product Closure with Low Extractables
Medical-Grade Lyophilization Closures Designed for Biologics, Vaccines, and High-Purity Pharmaceuticals in Type I Borosilicate Glass Vials
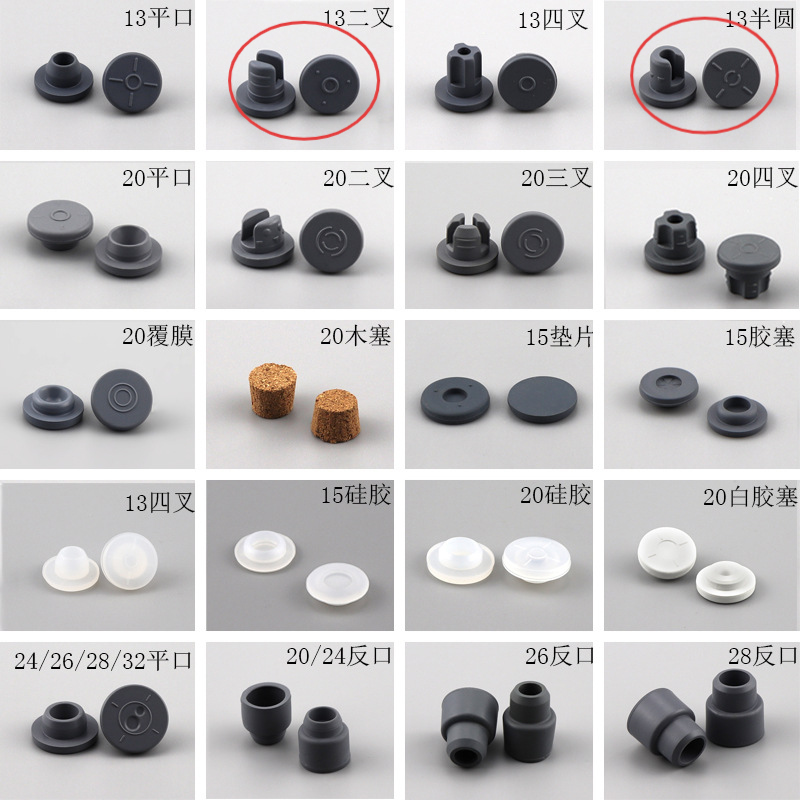
The 13mm, 15mm, and 20mm Butyl Rubber Stopper for Lyophilization from PharGlass is engineered specifically for freeze-dried (lyophilized) pharmaceuticals, offering superior moisture barrier performance, low extractables, and excellent resealability after vial sealing. These pharmaceutical lyophilization closures are ideal for biologic drugs, mRNA vaccines, monoclonal antibodies (mAbs), and sterile powder formulations that require long-term stability under controlled humidity conditions.
Unlike standard rubber stoppers, our lyophilization-grade butyl rubber stoppers are manufactured using high-purity raw materials and undergo rigorous extractables testing to ensure they meet the stringent requirements of USP <661>, EP 3.1.9, and FDA 21 CFR 177.1520 standards.
Manufactured under ISO 9001:2015 and GMP-compliant conditions in Henan, China, every stopper is tested for moisture permeability, visual defects, and bioburden control — ensuring consistent quality and regulatory compliance for global markets.
✅ Key Features:
| Feature | Description |
|---|---|
| Material | Medical-grade Butyl Rubber, Bromobutyl, or Chlorobutyl (recommended for freeze-dried applications) |
| Design Type | Flat-faced or dual-lip – optimized for vacuum sealing during lyophilization cycle |
| Moisture Barrier | Exceptional resistance to water vapor ingress – critical for dry powder stability |
| Low Extractables | Minimizes leachables that could compromise drug purity or safety |
| Customizable Sizes | Available in 13mm, 15mm, 20mm, 25mm, 28mm, 30mm, 32mm |
| Color Options | Gray, Red, White, Black, Blue, Yellow (customizable per request) |
| OEM/ODM Support | Yes – custom size, color, logo printing, surface treatment |
| Free Sample Policy | Free samples available for compatibility testing |
Technical Specifications: 13mm, 15mm & 20mm Lyophilization Rubber Stopper
| Parameter | Specification |
|---|---|
| Product Name | 13mm 15mm 20mm Butyl Rubber Stopper for Pharmaceutical Lyophilization |
| Model Number | 03 |
| Brand Name | PharGlass |
| Material | Butyl Rubber, Bromobutyl Rubber, Chlorobutyl Rubber |
| Color | Gray, Red, White, Black, Blue, Yellow, or Customer Request |
| Sizes Available | 13mm, 15mm, 20mm, 25mm, 28mm, 30mm, 32mm |
| Place of Origin | Henan, China |
| Usage | Primary closure for freeze-dried glass vials |
| Processing Service | Precision compression molding |
| Packaging | Plastic Bag + Export Carton |
| MOQ | 10,000 pcs |
| Sample Availability | Free Samples |
| OEM/ODM Support | Acceptable |
| Feature | Medical Grade, Lyophilization Grade, Low Moisture Permeability, Low Extractables |
| Single Package Size | 10×20×15 cm |
| Gross Weight | 1.000 kg per unit |
| Certifications | ISO 9001:2015, ISO 14001, OHSAS 18001 |
| Regulatory Compliance | USP <661>, EP 3.1.9, JP XVII, FDA 21 CFR 177.1520 |
Why Chlorobutyl Rubber Is the Gold Standard for Freeze-Dried Products
For lyophilized (freeze-dried) pharmaceuticals, the choice of rubber material is critical. Here’s why chlorobutyl rubber is preferred over standard butyl:
🔹 Superior Moisture Barrier: Chlorobutyl reduces water vapor transmission by up to 30% compared to butyl rubber (Source: Journal of Pharmaceutical Sciences, 2022).
🔹 Lower Extractables Profile: Essential for sensitive biologics where even trace impurities can affect efficacy.
🔹 Better Chemical Resistance: Resists degradation from residual solvents and excipients used in formulation.
🔹 Enhanced Seal Integrity: Maintains tight closure under vacuum conditions during the lyophilization cycle.
According to the European Pharmacopoeia (EP 3.1.9), halogenated butyl rubbers (e.g., chlorobutyl) are recommended for high-purity biologics due to their low leachables and high chemical inertness.
Top Applications in Pharma & Biotech
- ❄️ Lyophilized Biologics (e.g., insulin, interferons, growth factors)
- 💉 mRNA Vaccines (e.g., Pfizer, Moderna-style platforms)
- 🧬 Monoclonal Antibodies (mAbs) and Recombinant Proteins
- 🔬 Viral Vector Therapies and Gene Editing Tools
- 🧪 Clinical Trial Packaging & Reference Standards
Why Global Pharma Brands Trust PharGlass
As a leading OEM supplier of pharmaceutical closures, PharGlass provides end-to-end support including:
- Full Drug Master File (DMF) documentation
- Extractables & Leachables (E&L) study support
- COA, TDS, and batch traceability
- Custom tooling for unique geometries
- Fast prototyping (samples in 3–5 days)
- Global shipping with DDP/DAP options
All products are manufactured in a cleanroom environment and can be supplied pre-sterilized (gamma or autoclave).
📞 Request a Quote or Free Sample Today!
Email us at sales@pharglass.com or visit https://www.pharglass.com to explore our full range of pharmaceutical rubber stoppers, glass vials, and custom packaging solutions.


